I am working with the following hypothesis of the transition of fingers and phalanges:
I II III IV V
P-2-3-4-5-x Pterosaurx-x-2-3-4-x Basal Paraves (Scansoriopterygids)
P = pteroid. Numbers represent the number of phalanges. Roman numerals represent fingers (digits).
For example, this shows 5 phalanges in the fourth finger (IV) of pterosaur.
Summary of Changes:
The first step occurred in pterosaur with the loss of the fifth finger (V).
Then in the transition to basal paraves:
The pteroid was lost. (It continues as a prepollex in bird embryo).
The first finger (I) was lost. (It continues as a vestigial digit in bird embryo).
Each remaining digit lost one phalanx each.
In later Paraves the fourth finger (IV) was shortened.
Digit II becomes the alula.
This transition from pterosaur digits to the digits of a basal paraves like Scansoriopteryx, is consistent with all the known evidence. It is not contrary to any known evidence.
This transition from pterosaur digits to the digits of a basal paraves like Scansoriopteryx, is consistent with all the known evidence. It is not contrary to any known evidence.
In regard to the pteroid/prepollex, the change I am proposing is similar to the hexadactyl origin hypothesis (HOH).
In other respects it is like the pyramid reduction hypothesis for reduction in phalanges as well as lost digits.
Hexadactyl origin hypothesis (HOH)
https://vargaslab.files.wordpress.com/2009/03/vargasfallon2005b3.pdf
Good reference:
http://dml.cmnh.org/2005Dec/msg00213.html
SOCS2
https://www.academia.edu/1486957/Transcriptomic_analysis_of_avian_digits_reveals_conserved_and_derived_digit_identities_in_birds
Welten et al
Good description of pyramid reduction hypothesis even though they do not agree with it:
http://aracnologia.macn.gov.ar/st/biblio/Xu%20and%20Mackem%202013%20Tracing%20the%20Evolution%20of%20Avian%20Wing%20Digits.pdf
For more details see:
https://pterosaurnet.blogspot.com/2015/12/pyramid-reduction-hypothesis.html
http://pterosaurnet.blogspot.com/2014/07/pterosaur-fingers.html
https://pterosaurheresies.wordpress.com/2013/12/11/is-the-prepollex-radial-sesamoid-analogous-to-the-pteroid/
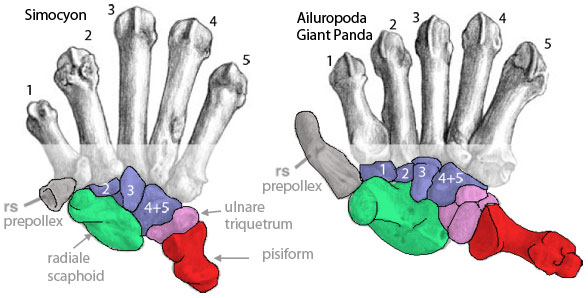
Other creatures with a prepollex:
In other respects it is like the pyramid reduction hypothesis for reduction in phalanges as well as lost digits.
Hexadactyl origin hypothesis (HOH)
https://vargaslab.files.wordpress.com/2009/03/vargasfallon2005b3.pdf
The early pantetrapod Devonian ancestors of
birds were polydactylous, with up to eight fingers
on both forelimbs and hindlimbs (Clack, 2002). In
the embryos of several modern tetrapods, it is
possible to observe the presence of mesenchymal
condensations other than those of digits 1-5 that
may represent vestiges of the additional digits of
early pan-tetrapods. A mesenchymal condensation
in front of digit 1 is called a prepollex, and a
mesenchymal condensation found posterior to
digit 5 is called a postminimus.
Although only three digits develop in the wing of
the chicken, recent work on the expression of the
Sox9 gene allowing the visualization of mesenchymal
condensations has revealed that six mesenchymal
condensations can be found in the
developing wing that can be compared to digital
condensations (Welten et al., 2005). Remarkably,
it is possible to interpret these condensations as
consistent with the proposal that the anteriormost
condensation may be a prepollex, the following
three digital condensations are digits 1, 2 and 3,
the next condensation is a vestige of digit 4, and
the posteriormost condensation (named ''element
X'' by Welten et al., 2005) can be assumed to be a
vestige of digit 5
In fact, there is no evidence of phalangeal initiation of any kind [no phalanges] in theThe bird prepollex has no phalanges, just like the pteroid which also has no phalanges!
brief Sox9 expressing region anterior to the first digit in the wing. It remains plausible that this is a prepollex
Good reference:
http://dml.cmnh.org/2005Dec/msg00213.html
In principle, hands of adult tetrapods where the first four conventional digits plus an ossified prepollex are present while the 5th finger is completely absent have been known for a long time.ALSO:
https://www.academia.edu/1486957/Transcriptomic_analysis_of_avian_digits_reveals_conserved_and_derived_digit_identities_in_birds
To discover genes that specifically contribute to the second and third wing digit identities, we performed differential expression analysis of the mRNA-seq data between samples LFb and LFc. We found two genes, Tbx3 and Socs2, with high expression in sample LFc (Supplementary Fig. 9 and Fig. 3a). To our knowledge no studies have been published indicating a role for Socs2 in limb development. ISH confirms its strong expression in the third forelimb digit to the exclusion of all other digits in forelimb and hindlimb(Fig.3b-g). Recently it has been shown that the third forelimb digit has a unique mode of development in birds8. This, combined with our gene expression survey,supports the idea that the third wing digit has a unique derived identity in birds.https://www.ncbi.nlm.nih.gov/pubmed/16419040
Suppressor of cytokine signaling (SOCS)-2 regulates normal postnatal growth and its deficiency in mice causes gigantism with increased bone length and proportional enlargement in skeletal muscles.http://www.jci.org/articles/view/22710
SOCS2 negatively regulates growth hormone action in vitro and in vivoMice deficient in SOCS2 display an excessive growth phenotype characterized by a 30-50% increase in mature body size. Here we show that the SOCS2-/- phenotype is dependent upon the presence of endogenous growth hormone (GH) and that treatment with exogenous GH induced excessive growth in mice lacking both endogenous GH and SOCS2. This was reflected in terms of overall body weight, body and bone lengths, and the weight of internal organs and tissues.http://www.tmd.ac.jp/artsci/biol/textbiodiv/17EvoDev7-18.pdf
Welten et al
The bird wing is of special interest to students of homology and avian evolution. Fossil and developmental data give conflicting indications of digit homology if a pentadactyl "archetype" is assumed. Morphological signs of a vestigial digit I are seen in bird embryos, but no digit-like structure develops in wild-type embryos. To examine the developmental mechanisms of digit loss, we studied the expression of the high-mobility group box containing Sox9 gene, and bone morphogenetic protein receptor 1b (bmpR-1b)-markers for precondensation and prechondrogenic cells, respectively. We find an elongated domain of Sox9 expression, but no bmpR-1b expression, anterior to digit II. We interpret this as a digit I domain that reaches precondensation, but not condensation or precartilage stages. It develops late, when the tissue in which it is lodged is being remodeled. We consider these findings in the light of previous Hoxd-11 misexpression studies. Together, they suggest that there is a digit I vestige in the wing that can be rescued and undergo development if posterior patterning cues are enhanced.
We observed Sox9 expression in the elusive ‘‘element X’’ that is sometimes stated to represent a sixth digit. Indeed, incongruity between digit domains and identities in theropods disappears if birds and other archosaurs are considered primitively polydactyl (more than 5 fingers). Our study provides the first gene expression evidence for at least five digital domains in the chick wing. The failure of the first to develop may be plausibly linked to attenuation of posterior signals.Pyramid reduction hypothesis
Good description of pyramid reduction hypothesis even though they do not agree with it:
http://aracnologia.macn.gov.ar/st/biblio/Xu%20and%20Mackem%202013%20Tracing%20the%20Evolution%20of%20Avian%20Wing%20Digits.pdf
One proposed mechanism postulates that an elevation in peripheral BMPs, signaling factors that modulate cell survival and proliferation [60,61], drove bilateral medial and lateral digital reduction [9]. This hypothesis is developmentally plausible, and is also consistent with the phalangeal reduction pattern seen in basal birds [9,23]. However, it predicts that the direct avian ancestor had a five-fingered hand with dominant digits II, III, and IV [9], which is inconsistent with the digital reduction data from basal theropods (e.g., all known basal theropods, including ceratosaurs, have a vestigial digit IV) [5,62–64]. In fact, the pyramid reduction hypothesis implies that either birds are not descended from theropod dinosaurs, or that some as yet to be discovered basal theropods were five-fingered with dominant digits II, III, and IV.
For more details see:
https://pterosaurnet.blogspot.com/2015/12/pyramid-reduction-hypothesis.html
http://pterosaurnet.blogspot.com/2014/07/pterosaur-fingers.html
https://pterosaurheresies.wordpress.com/2013/12/11/is-the-prepollex-radial-sesamoid-analogous-to-the-pteroid/
Other creatures with a prepollex:
